
The ADA enzyme deaminates adenosine to inosine, and it also deaminates 2′-deoxyadenosine (dATP) to deoxyinosine ( 2). The profound manner in which ADA deficiency causes immunologic dysfunction has been studied since its discovery in the 1970s ( 1). Collectively, these data implicate a B cell–intrinsic mechanism for alterations in B cell tolerance in the setting of partial ADA deficiency that is corrected by gene therapy.Ĭomplete adenosine deaminase (ADA) deficiency causes hepatic, skeletal, neurologic, and immunologic defects and results in failure to thrive, recurrent severe infections, and ultimately death if untreated. 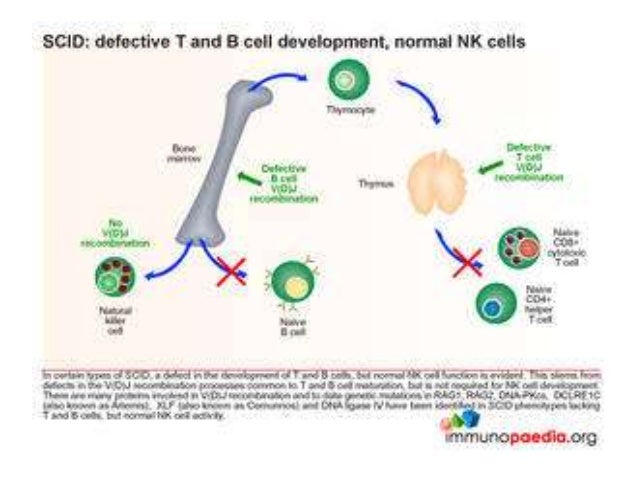
In vitro, agents that either block ADA or overexpress adenosine resulted in altered B cell receptor and TLR signaling. Remarkably, gene therapy with a retrovirus that expresses ADA resulted in the apparent correction of these defects, with normalization of peripheral B cell autoantibody frequencies. They demonstrate that ADA patients receiving enzyme replacement therapy had B cell tolerance checkpoint defects. In this issue of the JCI, Sauer and colleagues investigate the B cell defects in ADA-deficient patients. Adenosine deaminase (ADA) deficiency, a cause of X-linked severe combined immunodeficiency (SCID), is a case in point. It is paradoxical that immunodeficiency disorders are associated with autoimmunity. Hence, ADA plays an essential role in controlling autoreactive B cell counterselection by regulating BCR and TLR functions. Strikingly, after HSC-GT, ADA-SCID patients displayed quasi-normal early B cell tolerance checkpoints, as evidenced by restored removal of developing autoreactive and ANA-expressing B cells. We further observed impaired B cell receptor (BCR) and TLR functions in B cells after ADA inhibition, which may underlie the defects in B cell tolerance. We found that before HSC-GT, new emigrant/transitional and mature naive B cells from ADA-SCID patients contained more autoreactive and ANA-expressing clones, indicative of defective central and peripheral B cell tolerance checkpoints. To assess whether ADA deficiency affects the establishment of B cell tolerance, we tested the reactivity of recombinant antibodies isolated from single B cells of ADA-SCID patients before and after HSC-GT. However, autoimmune complications and autoantibody production, including anti-nuclear antibodies (ANAs), frequently occur in ADA-SCID patients after treatment. Restoration of purine metabolism and immune functions can be achieved by enzyme replacement therapy, or more effectively by bone marrow transplant or HSC gene therapy (HSC-GT). Adenosine deaminase (ADA) gene defects are among the most common causes of SCID.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
